Jewel Creation Education: stone setting, casting, engraving, polishing, soldering
Diamonds Education: Introduction, history, mining, cut types, diamond grading companies
How to Grade Diamonds: Color, clarity, carat, cut
Other How to Guides: How to clean diamond earrings, How to clean diamond rings
Stone and Diamond Cutting Education:
Stone cutting history, Stone cutting technique
Natural Gemstones Education: Gemstone history, what gemstones are,
Agate, Alexandrite, Axinite, Amethyst, Benitoite, Aquamarine, Cassiterite, Chrysoprase, Danburite, Diopside, Dioptase, Emerald, Labradorite, Moonstone, Sunstone, Garnet, Hambergite, Hematite, Jade jadeite and nephrite, Opal, Ruby, Sapphire, Turquoise, Topaz, Tourmaline, Vesuvianite
Precious Metals Education: Production and mining, as a metal, karats
 Gold being mined
Gold being minedThe richest and most heavily mined soils for the acquisition of gold are located in a relatively small part of the Southern part of Africa, such as Johannesburg. Nowadays goldmines are dug to a depth of up to several kilometers underground. For these reasons, Johannesburg is home to very advanced mining technology/equipment. Almost all of earth’s gold is hidden deep in its crust. The reason this area in South Africa is so rich in gold is because of a meteor hitting the earth there about 1 billion years ago, bringing much of the gold that was hidden underground in that area to the upper layers of the earth. It should be noted however that a substantial amount of the gold circulated today has been in circulation for hundreds and sometimes even thousands of years, as gold is often remelted into different shapes as fashion changes and older models are no longer wanted.

A gold nugget
The melting point of gold is 1063 РC / 1336,16 K. After gold cools down again to room temperature its mass shrinks by about 2%. It is very easy to take out of a mold for this reason.
A gold atom.
Another important chemical property of gold is that it is one of the best conductors. There are better conductors, but they are not as chemically non-reactive. For this reason pretty much every piece of electronics contains some gold. The average desktop computer contains around two grams of gold.
24 Carat gold is mostly used for gold trading and as a very early step in fine jewel production. Almost no jeweler uses 24 carat gold for jewels for several reasons. 24 carat gold (pure gold) is very hard to work with due to its brittle nature, making it hard to shape. However, assuming it is poured in a mold and require no physical modification, pure gold is still of little use in jewels because it is much more prone to break, acquire scratches and change shape from pressure.
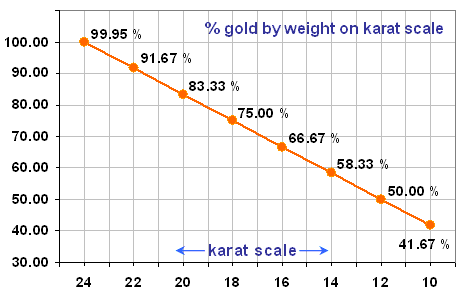
Gold carat scale
Considering longevity of each individual jewel is one of the biggest priorities of each jeweler, a mix of very strong metals are added to the mix, such as titanium. Most fine jewelers use 18k as a standard, because of its high gold purity yet strong nature. Gold jewels under 18k, such as one of the standards which is 14k is considered too low by fine jewelers. On the other hand, 22 which is the standard in many Asian countries, is considered not durable enough for long time wear. Many fine jewels are set with diamonds, only increasing the need for other strong metals to be to keep the valuable gem in place. In short, 18k gold ensures the high gold purity while maintaining durability for many years.
58.33% = 14k
75.00% = 18k
91.66% = 22k
95.83% = 23k
100% = 24k
A very interesting fact regarding gold carat is that white gold does not exist in a pure form. White gold is actually yellow gold, but a mixture of white metals causes it to appear white, differentiating it between yellow gold.
